AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Dalton atomic theory3/30/2024 
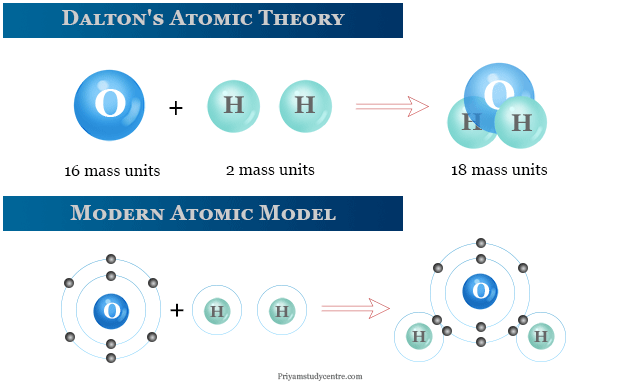
But atoms are the smallest particle to take part in a chemical reaction.Īccording to one postulate, the masses and densities of two different elements must differ. Like Isotopes of Hydrogen are deuterium and tritium, their masses are 1,2 and 3, respectively. But isotopes of the same element have different atomic masses. This proved that atoms were further divisible and were not the basic unit of matter.Īs one of the postulates states, the masses and densities of an element should be the same. These were protons, neutrons, and electrons. Later after the postulation of Dalton’s atomic theory, subatomic particles were discovered.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed